Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX)
CASRN 121-82-4 | DTXSID9024142
IRIS Toxicological Review of Hexahydro-1,3,5-Trinitro-1,3,5-Triazine (RDX) (Preliminary Assessment Materials)
On this page:
Alert
Notice - This site contains archived material(s)
Archive disclaimer
Archived files are provided for reference purposes only.
The file was current when produced, but is no longer maintained and may now be outdated.
Persons with disabilities having difficulty accessing archived files may contact the IRIS Webmaster for assistance.
Please use the contact us form if you need additional support.
Overview
In August 2013, EPA released the draft literature searches and associated search strategies, evidence tables, and exposure response arrays for RDX to obtain input from stakeholders and the public prior to developing the draft IRIS assessment. Specifically, EPA was interested in comments on the following:- Draft literature search strategies
- The approach for identifying studies
- The screening process for selecting pertinent studies
- The resulting list of pertinent studies
- Preliminary evidence tables
- The process for selecting studies to include in evidence tables
- The quality of the studies in the evidence tables
Background
Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX, or Royal Demolition eXplosive) is a military explosive with limited civilian uses. In the United States, RDX is produced at Army ammunition plants; it is not produced commercially.An oral reference dose (RfD) and an oral cancer slope factor are currently available on IRIS. A reassessment of the human health effects of RDX, including both cancer effects and effects other than cancer, is being undertaken.
Download(s)
This download(s) is distributed solely for the purpose of pre-dissemination peer review under applicable information quality guidelines. It has not been formally disseminated by EPA. It does not represent and should not be construed to represent any Agency determination or policy.
- Draft literature search and associated strategy and evidence tables for RDX (PDF) (93 pp, 1.8 MB, about PDF)
- IRIS December Public Science Meeting -- RDX Presentations
If you have a disability and the format of any material on our web pages interferes with your ability to access the information, please reach out to us using the Contact Us about IRIS form for assistance. To enable us to respond in a manner most helpful to you, please indicate the nature of the accessibility problem, the web address of the requested material, your preferred format in which you want to receive the material (electronic format (ASCII, etc.), standard print, large print, etc.), and your contact information.
Document Related Link(s)
- IRIS Toxicological Review of Hexahydro-1,3,5-Trinitro-1,3,5-Triazine (RDX) (Public Comment Draft)
- IRIS Toxicological Review of Hexahydro-1,3,5-Trinitro-1,3,5-Triazine (RDX) (Interagency Science Consultation Draft)
- IRIS Toxicological Review of Hexahydro-1,3,5-Trinitro-1,3,5-Triazine (RDX) (External Review Draft)
Federal Register Notices
Docket
Comments on the assessment may be submitted and reviewed using the Docket ID EPA-HQ-ORD-2013-0430Related Links
Critical Effect Systems
Tumor Sites
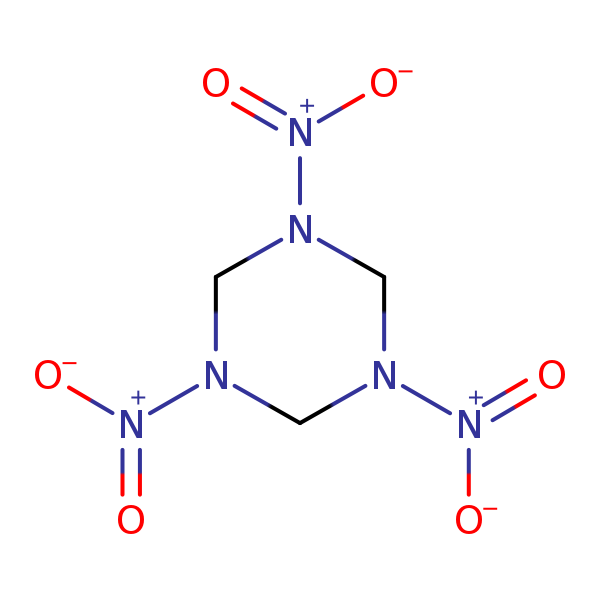
Chemical Structure for
Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX)
Synonyms
- Cyclonite
- Cyclotrimethylenenitramine
- Cyclotrimethylenetrinitramine
- Cyklonit
- Esaidro-1,3,5-trinitro-1,3,5-triazina
- Giekfol
- Heksoflen
- Heksogen
- Hexahydro-1,3,5-trinitro-1,3,5-triazin
- Hexahydro-1,3,5-trinitro-1,3,5-triazine
- Hexahydro-1,3,5-trinitro-s-triazine
- Hexogeen
- Hexogen
- Hexogen 5w
- Hexolite
- NSC 312447
- PBX(AF) 108
- PBX-MVF
- PBXN(AF) 108
- PBXW 108(e)
- RDX
- Research Development Explosive
- Royal Demolition eXplosive
- T4
- Trimethyleentrinitramine
- Trimethylenetrinitramine
- Trinitrocyclotrimethylene triamine
- UN 0072
- UN 0483
- perhydro-1,3,5-trinitro-1,3,5-triazine; RDX
- s-Triazine, hexahydro-1,3,5-trinitro-
- sym-Trimethylenetrinitramine
- trinitrotrimethylenetriamine
- 1,3,5-Triazine, hexahydro-1,3,5-trinitro-
- 1,3,5-Trinitro-1,3,5-triazacyclohexane
- 1,3,5-Trinitrohexahydro-s-triazine
- 1,3,5-triaza-1,3,5-trinitrocyclohexane
- 1,3,5-trinitro-1,3,5-triazinane
- 1,3,5-trinitrohexahydro-1,3,5-triazine
- 1,3,5-trinitroperhydro-1,3,5-triazine
- 121-82-4