1,4-Dichlorobenzene
CASRN 106-46-7 | DTXSID1020431
- IRIS Summary (PDF) (11 pp, 112.3 KB, about PDF)
IRIS Toxicological Review of Dichlorobenzenes (2004 External Review Draft)
On this page:
Alert
Notice - This site contains archived material(s)
Archive disclaimer
Archived files are provided for reference purposes only.
The file was current when produced, but is no longer maintained and may now be outdated.
Persons with disabilities having difficulty accessing archived files may contact the IRIS Webmaster for assistance.
Please use the contact us form if you need additional support.
Overview
The goal of this assessment is to evaluate both carcinogenic and non-cancer toxicity based on the proposed July 1999 Cancer Guidelines and other relevant guidance. This approach will provide a better understanding of the toxicity and will aid in making informed risk-based decisions for protection of human health. This assessment will be provided in the form of a Toxicological Review and IRIS summary sheets in the IRIS Database.Background
1,2-Dichlorobenzene is generally used as a chemical intermediate for making agricultural chemicals primarily herbicides, 1,3-dichlorobenzene is used as a fumigant in insecticides and 1,4-dichlorobenzene is used as a space deodorant, moth repellent, and as an intermediate in the synthesis of certain pesticides, dyes and resins. Chemical waste dump leachates and direct manufacturing effluents are reported to be the major source of pollution.Dichlorobenzene (DCB) was first evaluated by the Environmental Criteria and Assessment Office (ECAO), Office of Research and Development (ORD), Cincinnati, OH in 1987 for non-cancer and cancer endpoints. It was also evaluated by IRIS in 1996 (inhalation RfC; 1,4-DCB), 1992, 1990 (carcinogenicity and oral RfD respectively; 1,3-DCB), and 1991 (carcinogenicity and oral RfD; 1,2-DCB).
Status
This draft has been archived.Additional Information
EPA is hereby providing for public information a draft Toxicological Review, draft IRIS Summary, and charge to the external review panel for EPA's health assessment of 1,2-, 1,3-, and 1,4-dichlorobenzenes. These documents are provided for public viewing during an external scientific peer review period. While EPA is not soliciting public comments by this action, any scientific views received on the content of the Toxicological Review or IRIS Summary prior to 03/01/04 will be considered in subsequent drafts.Download(s)
This download(s) is distributed solely for the purpose of pre-dissemination peer review under applicable information quality guidelines. It has not been formally disseminated by EPA. It does not represent and should not be construed to represent any Agency determination or policy.
- Toxicological Review of Dichlorobenzenes (PDF) (146 pp, 7.6 MB, about PDF)
- Draft IRIS Summary: 1,2-Dichlorobenzene (PDF) (20 pp, 178.7 KB, about PDF)
- Draft IRIS Summary: 1,3-Dichlorobenzene (PDF) (15 pp, 155.3 KB, about PDF)
- Draft IRIS Summary: 1,4-Dichlorobenzene (PDF) (39 pp, 881.8 KB, about PDF)
- Charge to Reviewers, External Review Draft (PDF) (4 pp, 40.0 KB, about PDF)
- Consolidated Comments from the External Peer Review (Final) 2004 (PDF) (52 pp, 4.6 MB, about PDF)
- Consolidated Comments from the External Peer Review (1,4 - DCB Only) 2006 (PDF) (18 pp, 521.5 KB, about PDF)
If you have a disability and the format of any material on our web pages interferes with your ability to access the information, please reach out to us using the Contact Us about IRIS form for assistance. To enable us to respond in a manner most helpful to you, please indicate the nature of the accessibility problem, the web address of the requested material, your preferred format in which you want to receive the material (electronic format (ASCII, etc.), standard print, large print, etc.), and your contact information.
Document Related Link(s)
Federal Register Notices
Critical Effect Systems
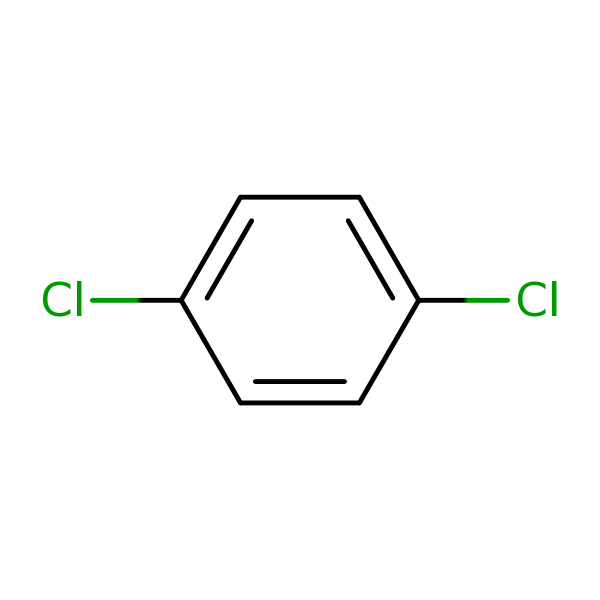
Chemical Structure for
1,4-Dichlorobenzene
Synonyms
- Benzene, 1,4-dichloro-
- Benzene, p-dichloro-
- Caswell No. 632
- Di-chloricide
- Dichlorobenzene
- Dichlorobenzene, para
- EPA Pesticide Chemical Code 061501
- Evola
- HSDB 523
- NCI-C54955
- NSC 36935
- PDB
- Paradi
- Paradichlorbenzol [German]
- Paradichlorobenzene
- Paradichlorobenzol
- Paradow
- Paramoth
- Parazene
- Persia-Perazol
- RCRA Waste Number u070
- RCRA Waste Number u072
- Santochlor
- UN 1592
- p-Chlorophenyl chloride
- p-Dichloorbenzeen [Dutch]
- p-Dichlorbenzol [German]
- p-Dichlorobenzene
- p-Dichlorobenzol
- p-Diclorobenceno [Spanish]
- p-Diclorobenzene [Italian]
- 1,4-Dichloorbenzeen [Dutch]
- 1,4-Diclorobenzene [Italian]
- 1,4-dichlorobenzene
- 106-46-7

