Hexachloroethane
CASRN 67-72-1 | DTXSID7020689
- Toxicological Review (PDF) (200 pp, 1.5 MB, about PDF)
- IRIS Summary (PDF) (34 pp, 300.8 KB, about PDF)
IRIS Toxicological Review of Hexachloroethane (Final Report)
On this page:
Overview
EPA has finalized the Toxicological Review of Hexachloroethane: in support of the Integrated Risk Information System (IRIS). Now final, this assessment may be used by EPA’s program and regional offices to inform decisions to protect human health.Background
The IRIS Toxicological Review of hexachloroethane provides scientific support and rationale for the hazard and dose-response assessment pertaining to chronic exposure to hexachloroethane. Hexachloroethane (C2Cl6) is a colorless, nonflammable, crystalline solid primarily used in the military for pyrotechnic devices. Hexachloroethane has been used as a polymer additive, moth repellant, plasticizer for cellulose esters, insecticide solvent, and for refining aluminum alloys. Hexachloroethane is not commercially distributed in the United States. Exposure to hexachloroethane is primarily occupational, via inhalation and dermal exposure during hexachloroethane manufacturing and metal refining. Exposure may occur in military or civilian personnel working with pyrotechnic devices containing hexachloroethane and via ingestion of contaminated drinking water or fish. Hexachloroethane was identified in Superfund sites.This assessment contains the derivation of a chronic oral reference dose (RfD), chronic inhalation reference concentration (RfC), and an oral cancer slope factor.
| Date | Description |
|---|---|
| Sep 1987 | The oral RfD and cancer assessment for hexachloroethane was posted to the IRIS database. |
| Apr 2010 | EPA initiated the interagency science consultation on the draft Toxicological Review of Hexachloroethane. |
| May 2010 | EPA released the External Review Draft for public review and comment. EPA also announced a public listening session to be held on June 16, 2010. [Federal Register May 13, 2010] |
| Aug 2010 | EPA announced an independent peer review meeting to be held in Arlington, VA on September 21, 2010. [Federal Register Aug 2, 2010] |
| Dec 2010 | EPA releases the comments from the public comment period and external review workshop. |
| Jul 2011 | EPA released the final toxicological review report, and summary report on the IRIS database. |
Download(s)
This document has been reviewed in accordance with U.S. Environmental Protection Agency policy and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
- IRIS Toxicological Review of Hexachloroethane (PDF) (200 pp, 1.7 MB, about PDF)
- IRIS Summary for Hexachloroethane (PDF) (34 pp, 270.5 KB, about PDF)
If you have a disability and the format of any material on our web pages interferes with your ability to access the information, please reach out to us using the Contact Us about IRIS form for assistance. To enable us to respond in a manner most helpful to you, please indicate the nature of the accessibility problem, the web address of the requested material, your preferred format in which you want to receive the material (electronic format (ASCII, etc.), standard print, large print, etc.), and your contact information.
Tumor Sites
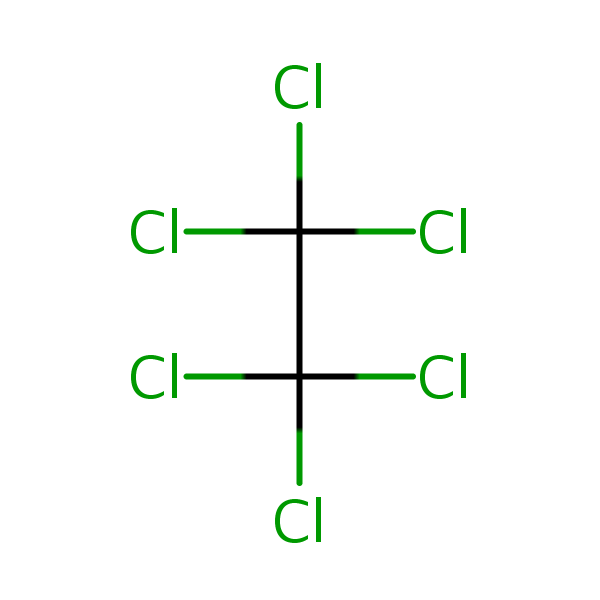
Chemical Structure for
Hexachloroethane
Synonyms
- Avlothane
- Carbon hexachloride
- Distokal
- Distopan
- Distopin
- Egitol
- Ethane hexachloride
- Ethylene hexachloride
- Falkitol
- Fasciolin
- Hexachlor-aethan
- Hexachloroethane
- Hexachloroethylene
- Mottenhexe
- NA 9037
- NCI-C04604
- Perchloroethane
- Phenohep
- RCRA Waste Number U131
- 1,1,1,2,2,2-Hexachloroethane
- 67-72-1