Ethylene oxide
CASRN 75-21-8 | DTXSID0020600
- Toxicological Review (PDF) (228 pp, 1.8 MB, about PDF)
- IRIS Executive Summary (PDF) (9 pp, 202.1 KB, about PDF)
- Background, History, and Other Supporting Documents (including Appendices)
Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (Final Report)
On this page:
Overview
EPA has finalized its Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide. This assessment addresses the potential carcinogenicity from long-term inhalation exposure to ethylene oxide. Now final, this assessment updates the carcinogenicity information in EPA’s 1985 Health Assessment Document. EPA’s program and regional offices may use this assessment to inform decisions to protect human health.Background
Ethylene oxide (EtO) is a gas at room temperature. It is manufactured from ethylene and is a chemical intermediate in the manufacturing of various chemicals. It is also used as a sterilizing agent for medical and dental equipment and as a fumigating agent for spices and certain other items. The highest levels of human exposure are in occupations involving contact with the gas in plants that manufacture or use EtO and in hospitals that sterilize medical equipment. EtO can also be inhaled by residents living near facilities that produce or use EtO or near sterilizing/fumigating facilities.The DNA-damaging properties of EtO have been studied since the 1940s. EtO is known to be mutagenic in a large number of living organisms, ranging from bacteriophage to mammals, and it also induces chromosome damage. It is carcinogenic in mice and rats, inducing tumors of the lymphohematopoietic system, brain, lung, connective tissue, uterus, and mammary gland. In humans employed in EtO-manufacturing facilities and in sterilizing facilities, there is strong evidence of an increased risk of cancer of the lymphohematopoietic system and of breast cancer in females.
| Date | Description |
|---|---|
| Jun 1985 | EPA published the Health Assessment Document for Ethylene Oxide (EPA/600/8-84/009F). |
| Sep 2006 | EPA released a draft reassessment for a 30-day public comment period. [Federal Register Notice September 22, 2006] |
| Jan 2007 | Peer review meeting of the Science Advisory Board (SAB) (public meeting). |
| Dec 2007 | EPA's SAB issued a final report on the review of the draft assessment of EtO. |
| Jul 2011 | EPA initiated Final Agency and an Interagency Science Discussion of a revised draft assessment of EtO. |
| Jun 2012 | EPA conducted further Agency review of a revised draft assessment of EtO. |
| Jul 2013 | EPA revised the draft assessment and released for additional public review and comment. The interagency science discussion draft of the assessment and related comments were also made available. [Federal Register Notice Jul 23, 2013] |
| Dec 2013 | EPA hosted an IRIS public science meeting to discuss the draft assessment released for public comment. |
| Aug 2014 | EPA submitted the draft assessment (revised in response to public comments) to the SAB for external peer review. [Federal Register Notice Aug 11, 2014] |
| Nov 2014 | EPA's SAB hosted a panel meeting for the SAB Chemical Assessment Advisory Committee (CAAC), augmented for the review of the draft IRIS assessment of EtO. |
| Aug 2015 | EPA's SAB issued a final report on its review of the draft assessment of EtO. |
| Oct 2016 | EPA submitted a revised draft for final Agency Review and Interagency Science Discussion. |
| Dec 2016 | EPA posted the final Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide to the IRIS database. |
Download(s)
This document has been reviewed in accordance with U.S. Environmental Protection Agency policy and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (EtO) (EPA/635/R-16/350Fa) (PDF) (228 pp, 1.8 MB, about PDF)
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (EtO) Appendices (EPA/635/R-16/350Fb) (PDF) (339 pp, 3.4 MB, about PDF)
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (EtO) Executive Summary (EPA/635/R-16/350Fc) (PDF) (9 pp, 206.1 KB, about PDF)
If you have a disability and the format of any material on our web pages interferes with your ability to access the information, please reach out to us using the Contact Us about IRIS form for assistance. To enable us to respond in a manner most helpful to you, please indicate the nature of the accessibility problem, the web address of the requested material, your preferred format in which you want to receive the material (electronic format (ASCII, etc.), standard print, large print, etc.), and your contact information.
Document Related Link(s)
- Evaluation of the Carcinogenicity of Ethylene Oxide (2006 External Review Draft)
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (Revised July 2013 External Review Draft) (Public Comment Draft)
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (2011 Interagency Science Discussion Draft)
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (2016 Interagency Science Discussion Draft)
- Evaluation of the Inhalation Carcinogenicity of Ethylene Oxide (Revised August 2014 External Review Draft) (SAB Review Draft)
Tumor Sites
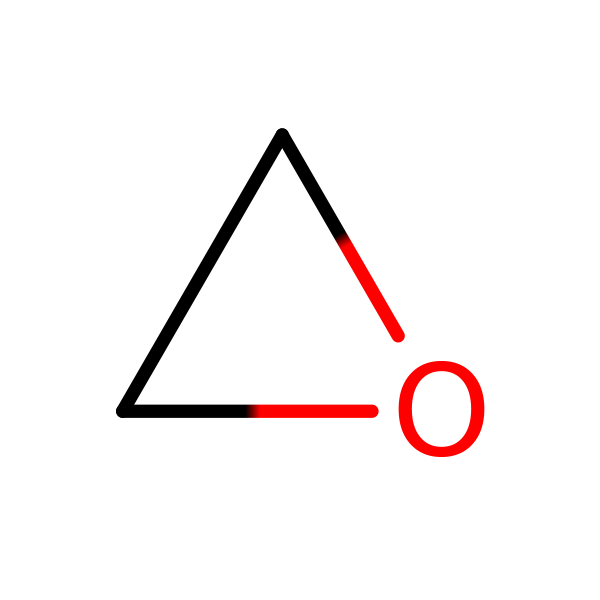
Chemical Structure for
Ethylene oxide
Synonyms
- AETHYLENOXID (GERMAN)
- AI3-26263
- ALPHA,BETA-OXIDOETHANE
- ANPROLENE
- Amprolene
- Anproline
- Caswell no 443
- DIHYDROOXIRENE
- DIMETHYLENE OXIDE
- E O
- ENT-26263
- EPA pesticide chemical code 042301
- ETO
- ETYLENU TLENEK (POLISH)
- Epoxyethane
- Ethene oxide
- Ethox
- Ethylene Oxide
- Fema no 2433
- NCI-C50088
- OXACYCLOPROPANE
- OXANE
- OXIDOETHANE
- OXIRAAN (DUTCH)
- OXIRAN
- OXIRANE
- OXIRENE, DIHYDRO-
- OXYFUME
- OXYFUME 12
- T-GAS
- USEPA/OPP Pesticide Code: 042301
- 1,2-EPOXYETHANE
- 75-21-8


